RYBREVANT® - Mechanism of Action Video
RYBREVANT® (amivantamab-vmjw) Mechanism of Action Video. For additional information, please refer to the Mechanism of Action scientific response
Johnson & Johnson

This summary explains the results of the PAPILLON study, which investigated a new potential treatment for patients with a type of lung cancer called non-small cell lung cancer (NSCLC) with EGFR exon 20 insertion mutations. These patients were treatment-naive or recently diagnosed and receiving amivantamab plus chemotherapy (carboplatin and pemetrexed) as a first line of therapy. Researchers evaluated the safety and effectiveness of a drug called amivantamab (brand name: RYBREVANT®) in combination with chemotherapy, comparing it with chemotherapy alone. The study included 308 patients from various countries, who were randomly given either the new combination treatment or just chemotherapy. Results from the study showed that patients who received amivantamab plus chemotherapy extended the time they lived without their cancer growing or spreading, and had more significant tumor shrinkage compared to those patients who received only chemotherapy. Serious adverse events were reported in 37% of the patients who received the combination compared to 31% of patients who received chemotherapy alone. The most common adverse reactions (≥20%) were rash, nail toxicity, stomatitis, infusion-related reaction, fatigue, edema, constipation, decreased appetite, nausea, COVID-19, diarrhea, and vomiting. Serious adverse reactions in ≥2% of patients included rash, pneumonia, interstitial lung disease (ILD), pulmonary embolism, vomiting, and COVID-19.
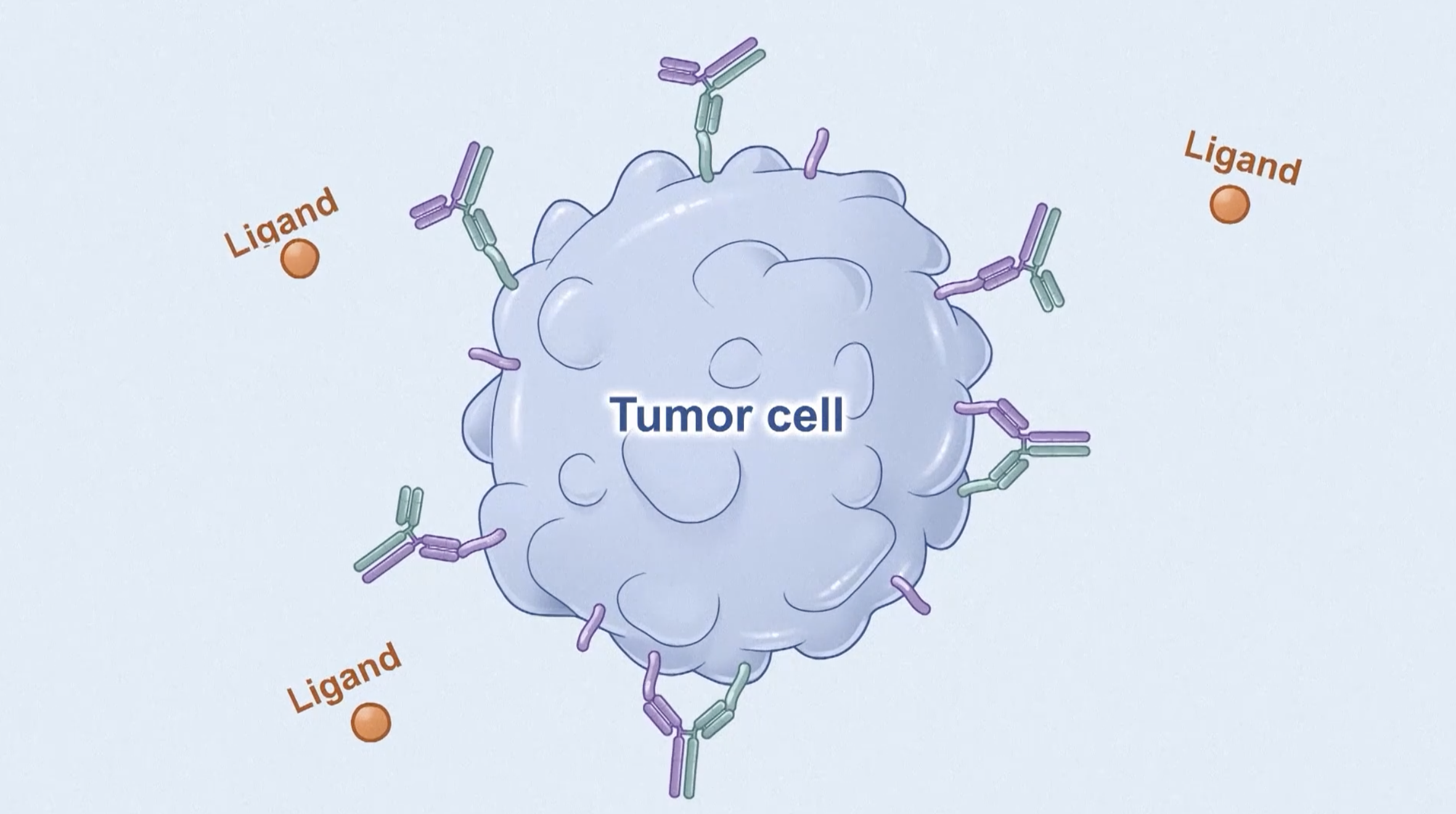
Understanding this new treatment is crucial because it offers a potential option for patients with advanced NSCLC with specific genetic mutations, which are hard to treat with existing therapies. Amivantamab, the primary component, targets the EGF and MET receptors or cancer cells, inhibiting their growth and spread, while chemotherapy directly kills the cancer cells. By identifying genetic mutations early on through testing, doctors can choose the most appropriate treatments right away, potentially extending the time patients can live without their cancer growing or spreading. The results from the PAPILLON study have led to the updated treatment guidelines recommending amivantamab plus chemotherapy as a preferred first-line option for patients with non-small lung cancer (NSCLC) with EGFR Exon 20 insertion mutations.
Key Takeaways:
Amivantamab plus chemotherapy extends the time without disease progression more effectively with 31% of patients living without having their tumors grow or spread at 18 months, compared to 3% of patients on chemotherapy alone. The median time for patients without their tumor growing or spreading was 11.4 months for amivantamab plus chemotherapy compared to 6.7 months with chemotherapy alone. Patients treated with the combination had a 60% lower risk of disease progression or death compared to those receiving only chemotherapy. More than 7 in 10 patients who received amivantamab plus chemotherapy had tumors that shrank or were no longer measurable compared to less than 5 in 10 patients who received chemotherapy alone. Serious adverse reactions occurred in 37% of the patients who received amivantamab plus chemotherapy compared with 31% of patients who received chemotherapy alone. The most common ARs (≥20%) were rash, nail toxicity, stomatitis, infusion-related reaction, fatigue, edema, constipation, decreased appetite, nausea, COVID-19, diarrhea, and vomiting.
The National Comprehensive Cancer Network Guidelines (NCCN Guidelines®) now recommends amivantamab plus chemotherapy as a preferred first-line treatment option for non-small cell lung cancer with EGFR Exon 20 insertion mutations.
Genetic Testing Importance: Early genetic testing for EGFR mutations can guide the selection of effective treatments and improve patient outcomes.
RYBREVANT® (amivantamab-vmjw) Mechanism of Action Video. For additional information, please refer to the Mechanism of Action scientific response
Results from the CHRYSALIS study in patients with non-small cell lung cancer with EGFR exon 20 insertion mutations who received amivantamab...
WARNINGS AND PRECAUTIONS
Warnings and Precautions for RYBREVANT® include infusion-related reactions, interstitial lung disease/pneumonitis, dermatologic adverse reactions, ocular toxicity, and embryo-fetal toxicity.
The safety population of RYBREVANT® with carboplatin and pemetrexed described in Warnings and Precautions was based on 151 patients in the PAPILLON study.