What is it about?
JADE DARE was a Phase 3 interventional clinical study designed to assess the efficacy and safety of abrocitinib 200 mg versus dupilumab 300 mg in adult patients with moderate-to-severe atopic dermatitis (AD). Patients (N=727) were randomly assigned to receive oral abrocitinib (200 mg per day) or subcutaneous dupilumab (300 mg every 2 weeks) for 26 weeks and were required to apply topical therapy to active lesions.
The primary endpoints were Peak Pruritus Numerical Rating Scale (PP-NRS)-4 response at week 2 and Eczema Area Severity Index (EASI)-90 at week 4. The key secondary endpoint was EASI-90 at week 16. PP-NRS4 response from day 15 to day 2, was also analysed using a sequential approach. Other secondary endpoints included the proportions of patients achieving EASI-90 and PP-NRS4 at all other scheduled timepoints. In addition, days free from medicated topical therapy, changes from baseline in Dermatology Life Quality Index and Patient-Oriented Eczema Measure, and the proportions of patients achieving the stringent endpoint of EASI-100 were assessed at all scheduled timepoints.
Details of these analyses and results of recent post-hoc analyses of JADE DARE are provided below.
Why is it important?
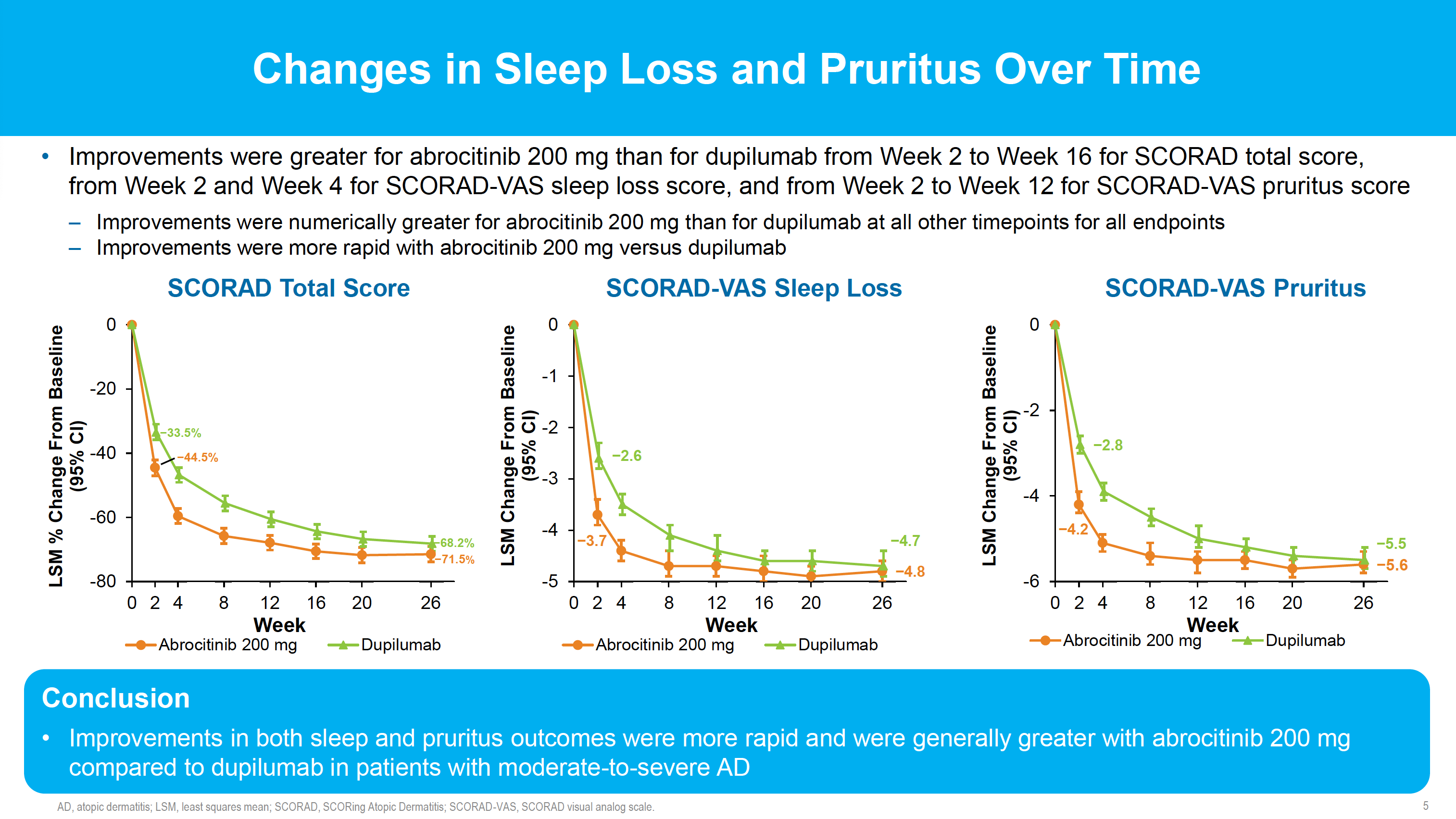
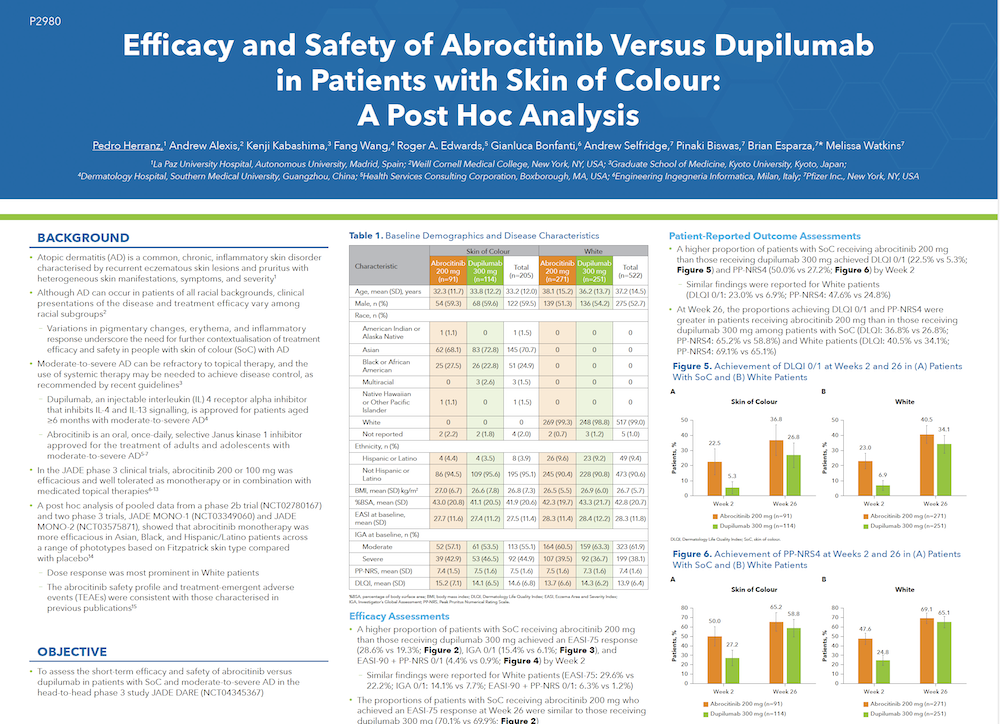
JADE DARE was the first head-to-head clinical study to assess the efficacy and safety of abrocitinib 200 mg versus dupilumab 300 mg in patients with moderate-to-severe AD. Significantly larger proportions of patients achieved the primary endpoint of PP-NRS4 at week 2 with abrocitinib (48%) than with dupilumab (26%), (p<0.0001). Similarly, a significantly greater proportion of patients achieved the additional primary endpoint of EASI-90 response at week 4 with abrocitinib compared with dupilumab, 29%, versus 15%, (p<0·0001). The proportion of patients who reached PP-NRS4 was higher in the abrocitinib group than in the dupilumab group at all timepoints from one day after the first dose to day 15.
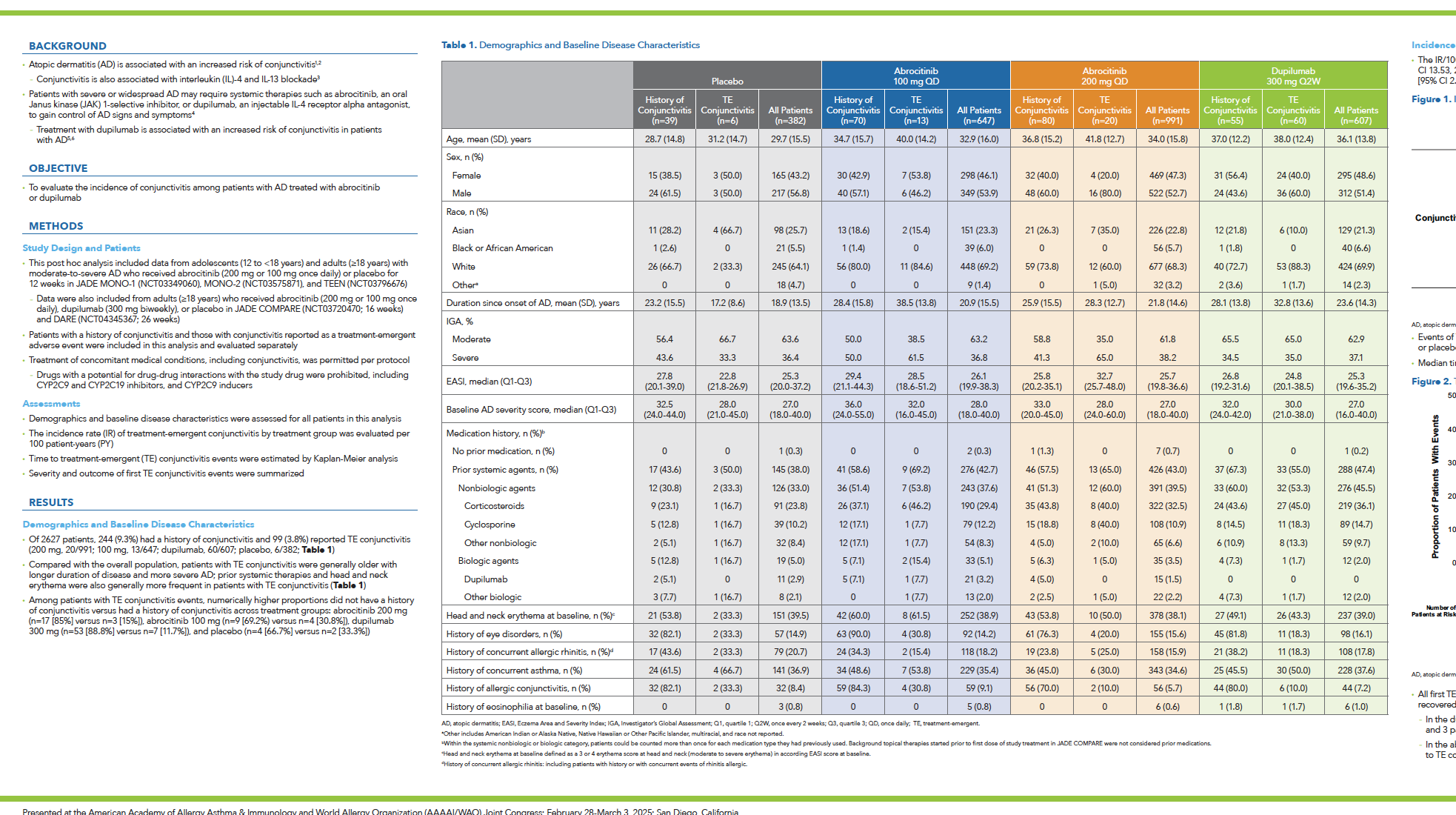
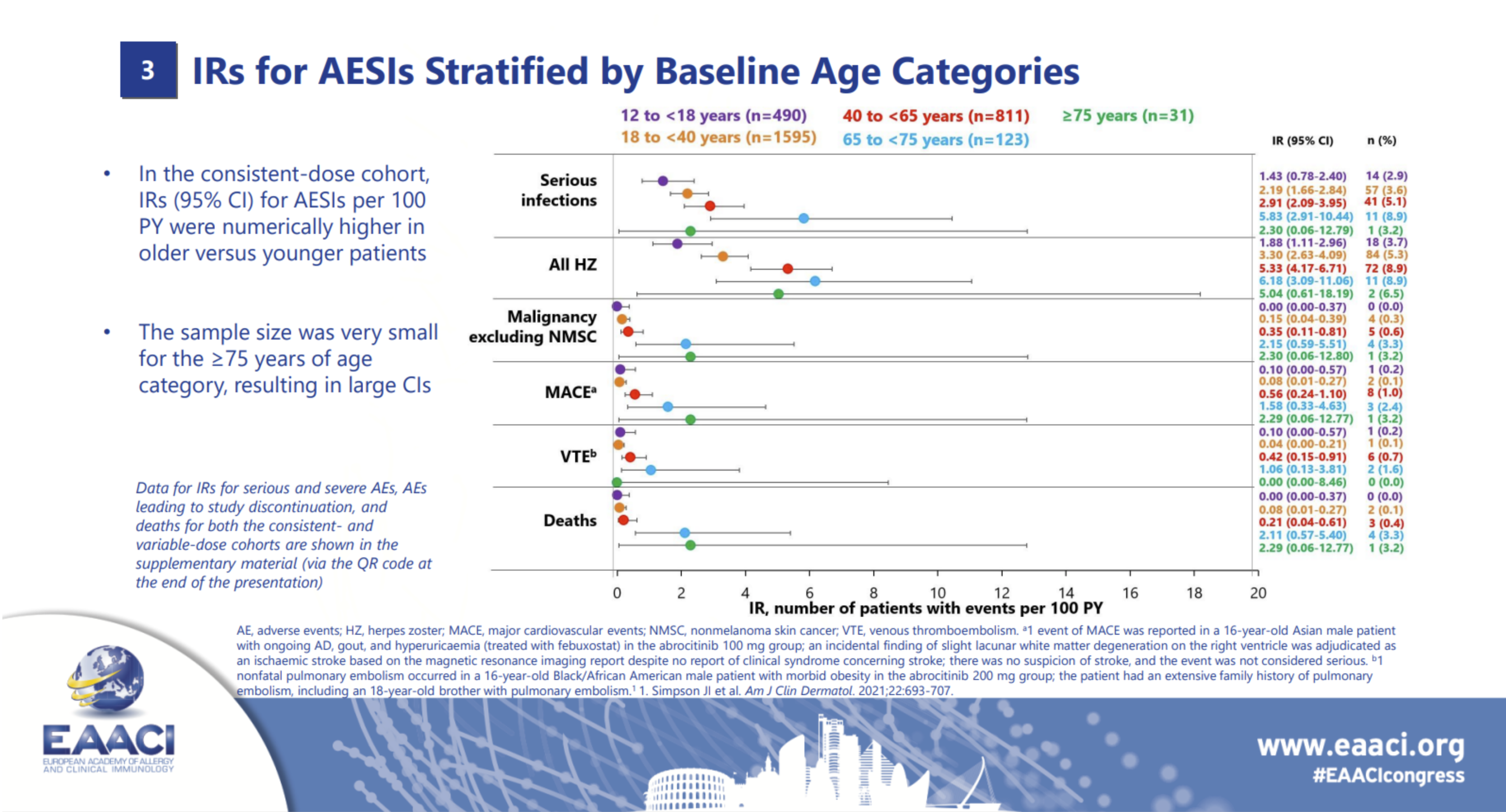
Among the 727 patients in the safety population, a total of 1,417 adverse events (AEs) were reported during the 26-week treatment period and the 28-day follow-up (817 with abrocitinib and 600 with dupilumab). A higher proportion of patients receiving abrocitinib experienced AEs compared to those receiving dupilumab (74% vs 65%). However, the rates of serious or severe AEs, as well as events leading to treatment discontinuation, were similar between both groups.
The authors stated that these “results suggest that oral abrocitinib 200 mg per day offers some advantages over dupilumab for patients with moderate-to-severe AD who require systemic treatment.”
EM-GLB-ARO-0141 | December 2025 Page published: 03-Feb-2026